PROJECT EYEMATICS
In Germany, around 10,000 new cases of blindness occur every year, which corresponds to an incidence of 12.3 per 100,000 inhabitants per year. Age-related macular degeneration alone accounts for 50% of blindness and diabetic retinopathy for 17%. The probability of suffering from one of these eye diseases increases significantly with age. As a result of increasing life expectancy, the number of blind or visually impaired people will also increase and the need for ophthalmic care will rise.
What these eye diseases have in common is that they can be treated with intravitreal injections (IVI). With intravitreal injections, a medication is administered directly into the vitreous body of the eye. The aim is to maintain or improve vision. Furthermore, IVOM is the most common medical procedure in Germany and is performed around 1,000,000 times a year.
The injections must be carried out several times. However, the strategies for the best possible treatment intervals have not yet been sufficiently investigated outside of clinical studies (i.e. in the “real world”). The situation within our consortium with regard to different treatment regimens is exemplary for Germany: at the university hospitals in Tübingen and Münster, IVOM treatments are generally planned in series of three consecutive injections according to the so-called pro-re-nata regimen, while in Greifswald and Aachen, some IVOM patients are treated according to the treat-and-extend regimen with single injections and individually adapted treatment intervals.
Initial practical evaluations have shown that treatment outcomes in Germany do not match the published good study results, while patient outcomes in other countries such as the UK and France appear to be significantly better. In Germany, there is a lack of multicenter data collection of real treatment data that would allow a detailed analysis and thus improved patient care.
The aim of the “EyeMatics” project is to make clinical data from patients with common eye diseases who receive IVI therapy accessible for scientific evaluation in order to improve patient care. EyeMatics combines individual data sources from various inpatient and outpatient care systems and makes this wealth of data accessible across all locations. As an example of meaningful data preparation and use, a clinical dashboard will be introduced at each site to support researchers in generating research questions by presenting the data from the data integration centers.
The overall objective of the project is to be realized by achieving the following sub-objectives:
- Provide for the first time a core data set for the exchange of medical patient data in ophthalmology to harmonize cross-site data collection and analysis,
- Demonstration of a functional, privacy-preserving infrastructure for the storage and exchange of data for this dataset,
- Integration of functional automated mechanisms for data extraction and merging,
- Setting up a dashboard as a data visualization and analysis platform,
- Routine recording and analysis of Patient Reported Outcomes (PRO) and integration of these results into the clinical dashboard,
- Development of AI tools to monitor and predict disease progression and treatment success based on the core dataset and OCT data and integration of these results into the clinical dashboard,
- Proof of an efficient rollout through the connection of two additional locations, another university hospital and a non-university tertiary care hospital, and
- Integration of the results into the statements of the German Ophthalmological Society.

Workpackages
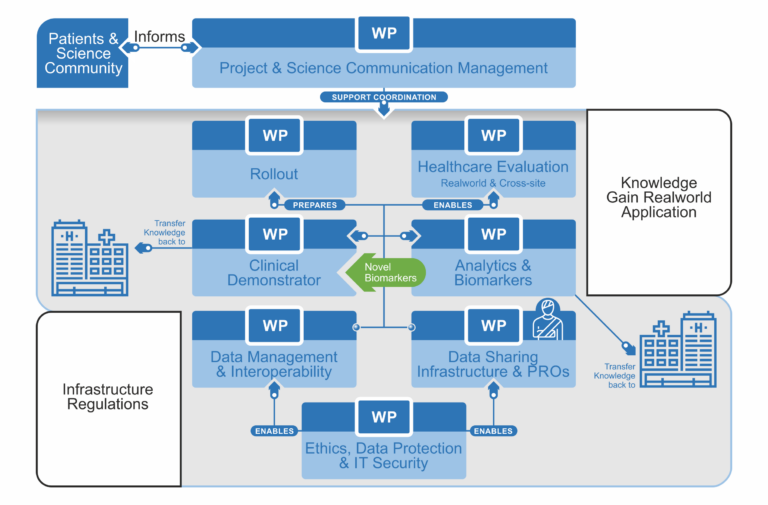
WP01 – Projectmanagement And Science Communication
This work package includes coordinating the work of all project partners involved and communicating with them. This work package includes the coordination of the work of all project partners involved and communication with them, the organization and implementation of regular project meetings, project documentation, reporting and public relations as well as communication with the project sponsor.
wP02 – Ethic, Data Protection and IT-Security
This work package includes the development and gradual adaptation of the data protection and IT security concept. A workflow for collecting broad consent, including additional modules, is to be established at all locations in the respective eye clinics. A vote by the responsible ethics committees and the Use and Access Committees (UACs) will be obtained to ensure that the project is carried out in a legally compliant manner.
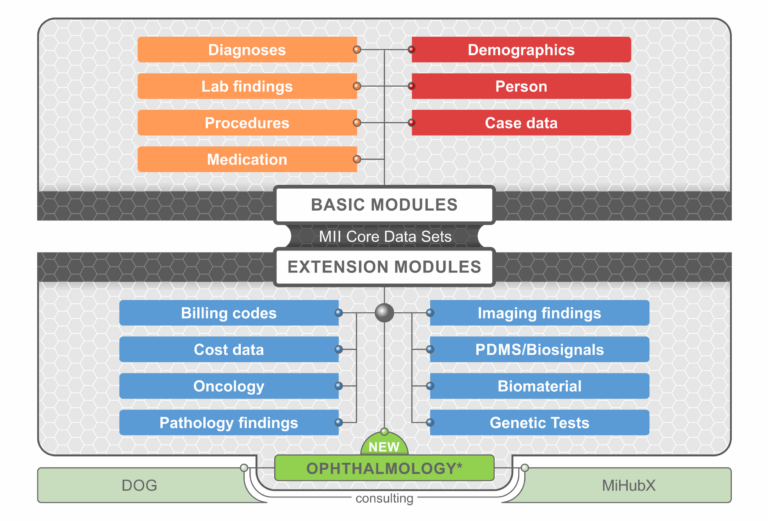
wP03 – Datenmanagement And Interoperatbilty
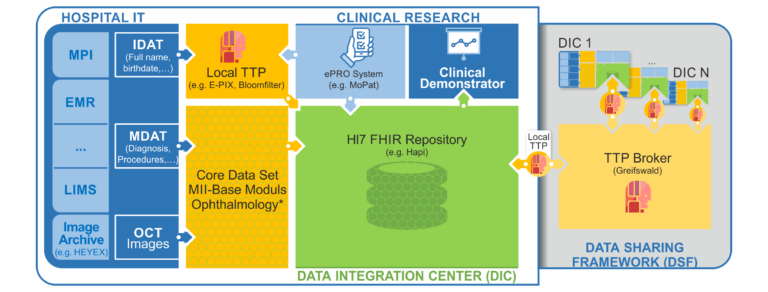
The project aims to develop a core dataset for ophthalmology as an MII extension module “Ophthalmology”. This forms the basis of the project and is designed in close coordination between the medical representatives of the clinics and the data integration centers. In addition, the expertise of the “Ophthalmology and Diabetes” use case of the Digital Innovation Hub MiHubX can be drawn upon. The content of the core dataset will relate to data from the MII basic modules (demographics, diagnosis, procedures, medication) and includes eye-specific data such as visual acuity, tonometry or refraction data as well as imaging metadata mainly from optical coherence tomography. Technically, the data set is implemented in FHIR.
An ETL pipeline will export the required medical elements from the hospital systems to a local FHIR repository at each DIC. This process requires a local TTP, which has already been set up in each DIZ during the first funding phase of the MII. In addition, image data metadata should be stored so that image queries can be carried out and image data can be requested if necessary.
wP04 – DATA EXCHANGE INFRASTRUCTURE AND PATIENT REPORTED OUTCOMES
The EyeMatics technical infrastructure provides for the local data integration centers to extract the core data sets from the primary hospital systems and store them in local FHIR repositories. EyeMatics will also collect Patient Reported Outcomes (PRO), which will be captured via an electronic system and also stored in the local FHIR repositories. A local TTP enables data set linkage.
The Data Sharing Framework (DSF) developed as part of the MII is to be used as the basis for exchanging data across the sites. A federated trust office pseudonymizes and links the data records so that data exchange across locations can be guaranteed in compliance with data protection regulations.
WP05 – CLINICAL DEMONSTRATOR
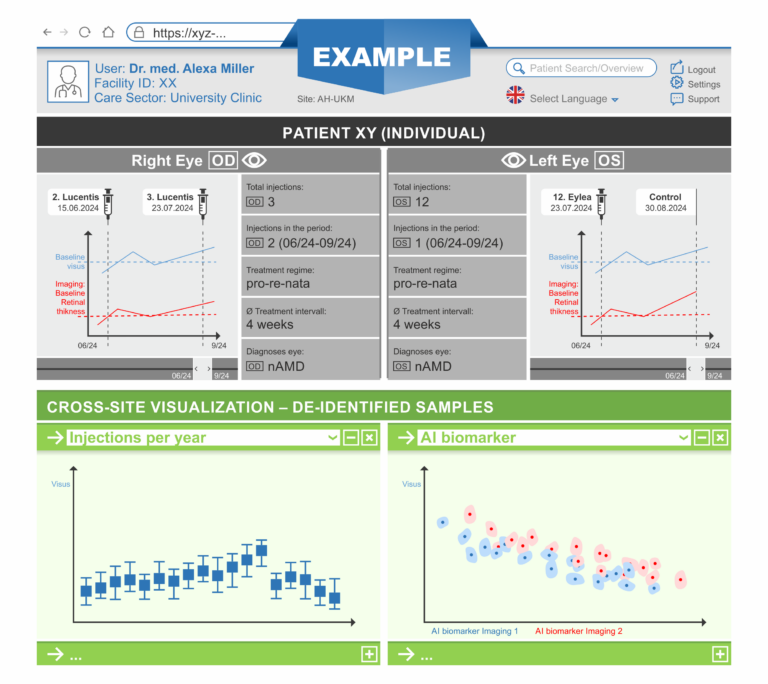
This work package consists of the development and implementation of a dashboard suitable for UC. An important aspect is the consideration and involvement of end users. Close cooperation and a feedback process with the users is essential. This includes in particular an evaluation of user-friendliness. Based on the efforts of WP03 and WP04, patient data available locally in the DIZ and aggregated data from other sites should be presented in a combined form. Due to the access and presentation of sensitive patient data, high demands on data security and access concepts must be met. This work package concludes with the distribution of the software solution developed here to the partners.
WP06 – AnalytiC and Biomarker
The aim of this work package is to develop AI tools for monitoring and predicting disease progression and treatment response based on the core dataset and raw data from optical coherence tomography (OCT), the main imaging modality used in IVOM treatment. This includes patient-level modeling across clinical sites. The resulting models will be used for the discovery of biomarkers indicative of treatment success. We will start with locally available data and add data from different sites via the AP04-based infrastructure. Site-specific differences will be investigated.
WP07 – Rollout
This work package is intended to demonstrate the transferability of the infrastructure developed in the project. The rollout partners will install the developed applications and establish the developed processes. This includes both the recruitment of patients via broad consent and the functioning ETL pipeline with data exchange connection.
WP08 – Evaluation OF HEALTH CARE
The aim of this work package is to develop a customized questionnaire to measure Patient Reported Outcomes (PROMs) that meets the needs of low vision patients undergoing IVOM therapy and the medical informatics infrastructure. Treatment quality indicators (treatment latency, frequency of monitoring and treatment, treatment adherence and consistency, medical outcomes) will be monitored during the study period both in the initial clinics and after the EyeMatics roll-out.
